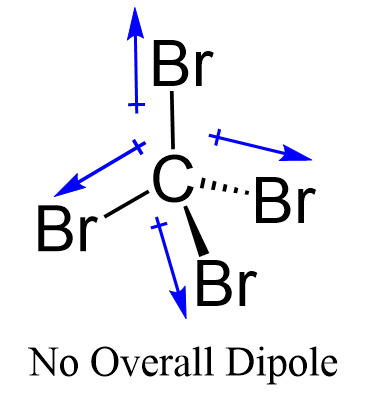
Provide the following information for the compound CBr4. a. number of shared electron pairs b. number of unshared electron pairs c. shape of molecule d. ionic, polar covalent, or nonpolar covalent

Is CBr4 Polar or Non-polar? (Carbon Tetrabromide) | Is CBr4 Polar or Non- polar? (Carbon Tetrabromide) Do you want to find out if Carbon Tetrabromide is a polar or nonpolar molecule? If yes

For the molecule CBr4, determine the following. a. name b. molecular geometry c. polarity d. type of intermolecular forces | Homework.Study.com

Draw the Lewis structure for CBr4 and provide the following information. a. number of electron groups b. electron pair geometry c. bond angle d. number of bonded electrons e. molecular geometry f.

CBr4 Lewis Structure (Carbon Tetrabromide) | CBr4 Lewis Structure (Carbon Tetrabromide) Hello everyone, we are back with yet another video on Lewis Structure. And this time, we are going to help...

A Both CCl4 and CBr4 are nonpolar. This means that London dispersion forces hold the molecules together in the liquid state. To boil the substances, the. - ppt download

Is CBr4 Polar or Nonpolar? - Polarity of Carbon tetrabromide | Molecular geometry, Covalent bonding, Polar

b. complete the following table. tell if the molecule is polar or nonpolar, draw the lewis structure for - brainly.com